Published by Lise Peeters, PhD on Mar 28, 2017 11:30:00 AM
Exosomes are cell-derived vesicles originating from multi-vesicular bodies and found in biological fluids such as blood, saliva, urine, and breast milk. Sizes of these extracellular vesicles (EVs) range between 30-100 nm. Due to their capacity to transfer proteins, lipids and nucleic acids, exosomes can influence various physiological and pathological functions (Yañez-Mo et al., 2015).
Exosomes play a key role in cell-cell communication and circulate in bloodstream, and therefore, are implicated as a disease biomarker for cancer and immune system disorders. However, there is limited information regarding efficient methods for obtaining pure exosomes. MBL offers a unique exosome purification kit that can help researchers purify exosomes from their sample.
Exosome purification
The current problem impeding progress in EV research is the lack of a standardized purification method. The disadvantages of ultracentrigugation (UC), filtration, and precipitation can be overcome by making use of affinity purification. Most of the immunoaffinity purification protocols used for the isolation of EVs rely on antibodies against common exosome protein markers, like tetraspanins (CD9, CD63, CD81) and tumor associated markers (EpCAM) for isolation and analysis of tumor-derived EVs directly from human plasma. Magnetic beads, for example, coated with antibodies against common EV protein markers may facilitate the purification of homogenous EV subpopulations present in biofluids. These specific EV populations, which can be obtained by immunoaffinity-based purification, can be desired in some applications, like cancer diagnostics.
How can ExoCapTM help you?
ExoCapTM-SP kits have been optimized for serum, plasma and cell culture supernatant, but these kits can also be used for urine and cerebrospinal fluid (CSF) (Junker et al., 2016). ExoCapTM-SP kits use functionalized MagnosphereTM magnetic particles for exosome immune precipitation. The beads are coupled to antibodies that recognize exosome surface antigens like tetraspanins (CD9, CD63, CD81) and tumor associated markers (EpCAM).
ExoCapTM Streptavidin Kits are designed for customized isolation and analysis of exosomes. Coupling of the biotinylated antibody with the magnetic streptavidin bead allows exosome isolation for your target of interest (Figure 1).
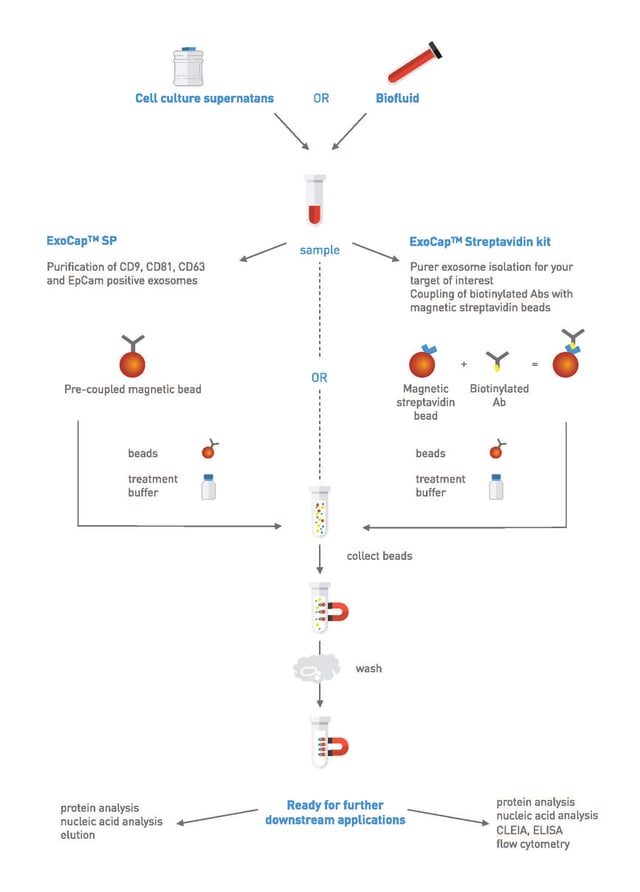
Figure 1: Schematic presentation of ExoCapTM and ExoCapTM Streptavidin Kit
ExoCap™ kit and optimized protocols for downstream applications
Both ExoCap™-SP and ExoCap™ Streptavidin kits contain a treatment buffer and a washing buffer, and respectively pre-coupled beads or streptavidin beads. The kits differ in the fact that the ExoCap™-SP kit also contains an elution buffer. In addition, both kits have optimized protocols for downstream applications such as protein analysis, nucleic acid analysis or flow cytometry (Table 1). Detailed protocols on these downstream applications are included in the kits.
| ExoCap™-SP | ExoCap™ Streptavidin kit |
| with optimized protocols for: | with optimized protocols for: |
|
|
Table 1: ExoCap™ kit components and overview of its downstream applications
How important is pure isolation for your research?
Given the importance of exosomes in normal physiology and disease pathogenesis, more research is focusing on the employment of utilizing these vesicles as innovative biomarkers in the diagnosis of cancer, prognosis, and management of cardiovascular diseases (De Toro et al., 2015). Nowadays, cancer diagnostics rely on biopsies. The potential use of exosomes has the advantage of being a sensitive and non-invasive method, allowing the detection of tumors at an early stage. It has been reported that cancer exosomes have been found to be useful for the detection of many types of tumors: prostate, breast, ovarian cancer, glioblastoma, melanoma ... (De Toro et al., 2015). For these purposes, the purity of exosomes is of most importance. In this case ExoCap™ kits can offer a user-friendly, fast and high purity capture of exosomes.
References
Yanez-Mo M. et al. , Biological properties of extracellular vesicles and their physiological functions, Journal of Extracellular Vesicles, (2015) 4, 27066, doi.org/10.3402/jev.v4.27066
Junker K., Heinzelmann J., Beckham C., Ochiya T., Jenster G., Extracellular Vesicles and Their Role in Urologic Malignancies, European urology, (2016), 323-331, doi.org/10.1016/j.eururo.2016.02.046
De Toro J., Herschlik L., Waldner C. and Mongini C., Emerging roles of exosomes in normal and pathological conditions: new insights for diagnosis and therapeutic applications, Frontiers in Immunology, (2015) 6, 203, doi: 10.3389/fimmu.2015.00203