Published by Cheryl A. Guyre, Ph.D. on
Please Note: MBL International will be shutting down its operations effective December 31, 2024. Distribution of MBL products in the United States will be transferred to Cosmo Bio US on January 1st while European Distributors will remain unchanged. For any US inquiries regarding orders or support during this transition, reach out to Cosmo Bio: https://www.cosmobiousa.com/. For Non-US inquiries reach out to MBL in Japan: https://www.mblbio.com/.
Published by Cheryl A. Guyre, Ph.D. on Aug 26, 2015 3:39:00 PM

You’re sitting at your flow cytometer, staring at your CD8 x tetramer plot in anticipation of a tiny but meaningful group of dots appearing in the upper right quadrant. You hold your breath, click “acquire,” and watch… Wait, what? What’s this ugly diagonal?? How come ALL the CD8 positives are dual positive??! Hey, this is my control mouse! That one should be completely tetramer negative! Yep, I’ve been there too. Classic case of using a CD8 antibody that doesn’t play nice with tetramers.
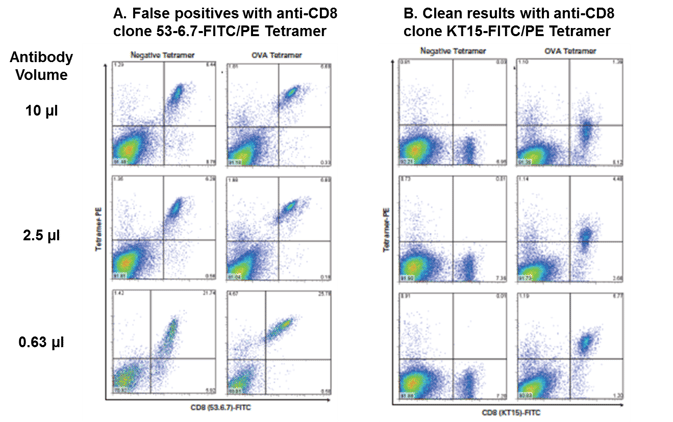
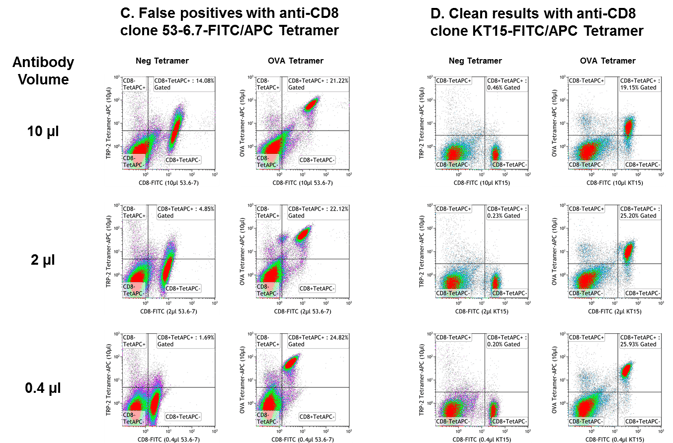
Certain anti-murine CD8 clones have been shown to affect tetramer staining1-6. In Current Protocols in Immunology, Altman and Davis caution, “Some MHC tetramers (e.g., H-2 Kb) have been observed to stain all CD8+ T cells, presumably via an interaction between CD8 and the α3 domain of the MHC molecule. In these cases, it is useful to perform a cross-titration series with the MHC tetramer and several distinct anti-CD8 antibodies, looking for CD8 antibodies which block with CD8-mediated interaction, but not the antigen-specific interaction. For example, Kb tetramers bind to all CD8+ T cells in the presence of the anti-CD8 antibody 53-6.71.”
We tested two different anti-murine CD8 FITC clones used to stain OT-I transgenic splenocytes with Negative (β-gal, A-B; TRP2, C-D) or OVA-specific (A-D) H-2 Kb tetramer. The use of anti-murine CD8 clone KT15 (B, D) eliminates confusing diagonals resulting from non-specific binding seen with clone 53-6.7 (A, C) in tetramer assays. PE-labeled tetramers (A, B) and APC-labeled tetramers (C, D) are shown. Titration of anti-murine CD8 antibody is recommended for optimized staining.
Selected References
